Insights From the CHRONIC ITN Final Workshop
Jacqueline Hilgendorf, University of Aveiro; Shankari Anna Balan, UK Centre for Ecology & Hydrology; Luca Boldrini, Belgian Nuclear Research Center; Nico van den Brink, Wageningen University; Wing Sze Chan, Roskilde University; Oihane Del Puerto, Syngenta Crop Protection AG; Valery E. Forbes, Florida Atlantic University; Colette Gaillard; Jian Ge, Aarhus University; Harmony Lautrette-Quinveros, Lunds University; Susanna Loureiro, University of Aveiro; Kevin J. Noort, UK Centre for Ecology & Hydrology; Sofie B. Rasmussen, Leiden University; Shivani Ronanki, Wageningen University; Martina Santobuono and Henriette Selck, Roskilde University; Matteo Schiavinato, Wageningen University; David Spurgeon, UK Centre for Ecology & Hydrology; and Martina Vijver, Leiden University
What Is CHRONIC?
The Marie-Curie Horizon 2020 International Training Network (ITN), CHRONIC (Chronic exposure scenarios driving environmental risks of chemicals), was established in 2020 to explore how chemicals that exert chronic effects through specific modes of action (MoA) and multigenerational effects interact with other environmental stressors to affect terrestrial and aquatic ecosystems. In this context, we examined the application of non-conventional endpoints that may provide greater mechanistic understanding and offer higher sensitivity to chemical–biota interactions that would otherwise remain undetected when relying solely on conventional apical endpoints. In partnership with SETAC, CHRONIC held its final workshop from 1-3 October 2024 in Brussels. The workshop brought together the CHRONIC Ph.D. student cohort and supervisors with external stakeholders representing business, government, academia and NGOs. The Ph.D. fellows presented their latest findings and reflections on their professional journeys. In addition, there were presentations by senior researchers, a stakeholder panel, breakout discussions and other interactive sessions.
Identifying the Challenge
Currently, the environmental risk assessment (ERA) of chemicals predominantly relies on measuring effects on conventional, standardized endpoints, such as growth, reproduction and mortality. These measures provide a consistent framework that ensures the reliability, comparability and reproducibility of ERA testing. Despite the intense testing of chemicals, pollution is acknowledged as one of the three main aspects of the triple planetary threat, alongside climate change and habitat/biodiversity loss. The impacts of chemical pollution are due, in part, to the inadequate assessment of low-level, chronic exposures and the use of test designs that are not well-suited for emerging pollutants. The CHRONIC project aimed to address these challenges of low-level chronic exposure by raising awareness, developing novel test methods, measuring non-conventional endpoints and assessing the added value of integrating these endpoints into ERA frameworks using adverse outcome pathways (AOPs).
Why Non-Conventional Endpoints?
Non-conventional endpoints (i.e., those not described by standardized guidelines and that extend beyond conventional, standardized endpoints) encompass measures of biological effect not commonly employed in standardized guidelines. These endpoints can provide insight into molecular and cellular mechanisms that typically act upstream of conventional endpoints (e.g., biochemical responses linked to the toxicokinetic and toxicodynamic characteristics of the chemical). Moreover, non-conventional endpoints which operate effectively at the individual level (e.g., locomotion or exploratory behavior) have been proven to be often more sensitive than conventional endpoints. Additionally, these individual effects can be plausibly linked to population-level effects, sometimes even revealing unexpected population-level effects on responses linked to mating, predator interactions, territoriality, etc., that would otherwise go undetected with current assessment methodologies. By comparing effect concentrations of conventional and non-conventional endpoints, we can improve our understanding of their relative sensitivity and inter-relationship and how this may impact, for instance, the safety factors used in risk assessment.
Within the CHRONIC project, researchers developed novel methods for non-conventional endpoints on several biological levels. This included, on the molecular/cellular levels, telomere shortening, serotonin concentrations, in vivo cytochrome P450 activity and epigenetic responses. At the individual level, behavioral endpoints, including chemical avoidance, feeding and burrowing, were assessed, as were predator-prey interactions. A 2025 review paper by Jacqueline Hilgendorf and colleagues compared conventional and non-conventional endpoints and demonstrated that the latter were generally more sensitive, being on average 56 times more sensitive than mortality, eight times more sensitive than reproduction, and twice as sensitive as growth. In 10% of the described cases, the NCEs were even more than 1000 times more sensitive than the CE. In 13%, NCEs showed effects when CEs were unaffected, hinting towards undetected population-level effects. These results were also underlined by the laboratory studies conducted through CHRONIC. Although measuring some non-conventional endpoints (e.g., gene expression analysis, predator-prey interactions) might currently be more labor-intensive to measure and analyze than conventional methods, they do provide information that leads to a deeper understanding of the hidden effects of chemical MoAs. This becomes increasingly important, especially now, as ECHA has introduced the emerging hazards neurotoxicity, immunotoxicity, and metabolic toxicity into regulatory assessment.
More Work Is Needed
However, for ERA, more work is needed to implement non-conventional endpoints, such as behavioral alterations. As supported in the CHRONIC consortium, Marlene Ågerstrand and colleagues argued in their 2025 paper that behavioral endpoints — such as changes in feeding, social interactions or avoidance — present a valuable complement to conventional ecotoxicological measures. In regulatory practice under the REACH Regulation or other EU frameworks, behavioral studies can generally be considered as supportive evidence, though rarely as the sole basis for risk assessment. The paper reports six documented cases — such as avoidance behavior in fish or altered foraging in bees — where behavioral endpoints contributed to regulatory decision-making, even if often given lower “weight” than standard endpoints like mortality or reproduction. These could serve as a blueprint for leveraging the information we have collected within the CHRONIC consortium.
Using these non-conventional endpoints under chronic low-dose exposure and/or in a multigenerational or multi-stressor context (e.g., with temperature changes or in chemical mixtures) further increases the environmental relevance of ERA practices.
AOPs Are the Ultimate Game-Changer
One major aim of CHRONIC was to characterize toxicant MoAs within an AOP, a conceptual framework that facilitates mechanistic understanding across different levels of biological organization. Especially with the introduction of the new CLP hazard class endocrine disruption, it becomes increasingly important for regulatory decision-making to understand the MoA of a substance, measure adverse outcomes at the organ, individual and population levels, and create a biologically plausible link between the two. AOPs provide a helpful conceptual framework to organize chemical effects data from a key initiating event and linking them to effects across levels of organization (up to populations) using key event relationships. This approach promotes the identification of pathways and any relevant gaps in knowledge, leading to hypothesis-driven science, targeted experimental designs and, eventually, improved mechanistic understanding. The goal of the AOP framework is to improve the predictive power of endpoints tested in ERA to population-level effects relevant to ecological protection goals. In turn, such mechanistic understanding will inform existing AOPs as a step toward reducing the number of tests needed in ERA practices.
The AOP principles are already well established for key human toxicological events and are published on www.AOPwiki.org, with explanations provided in the OECD Guidance Document on Developing and Assessing Adverse Outcome Pathways. Further research is necessary to determine causal relationships between key events and adverse outcomes at the population level. This can be achieved by generating quantitative AOPs. By including endpoints related to interspecies relationships, such as prey-predator interactions or those linked to species' functional roles in ecosystems (e.g., burrowing activity), we can further enhance the ecological relevance of these AOPs at population and ecosystem levels. Given the intrinsic variability of non-conventional endpoints, demonstrating that changes in them are consistently and robustly linked to impacts on survival, growth and reproduction – and ultimately population dynamics – will be essential.
The Solution: Integrating Non-Conventional Endpoints With AOPs by Aligning Them With a Chemical's MoA
Grouping chemicals by their MoAs can help quantify the impacts of low-level chronic exposures. The benefit of categorizing chemicals by MoAs is that it enables the identification of underlying mechanisms and of how these biochemical and physiological pathways are shared across different species. Developing an MoA-specific AOP framework (Figure 1) may allow us to better link effects on non-conventional endpoints to population-level effects and improve the precision of chemical hazard and risk assessment as it detects potential toxic effects at an early stage of the screening process. By incorporating appropriate feedback loops and compensatory processes into the AOPs, such as those that maintain cellular or physiological homeostasis, it should be possible to distinguish adverse effects from responses within the normal operating range of the exposed biological systems.
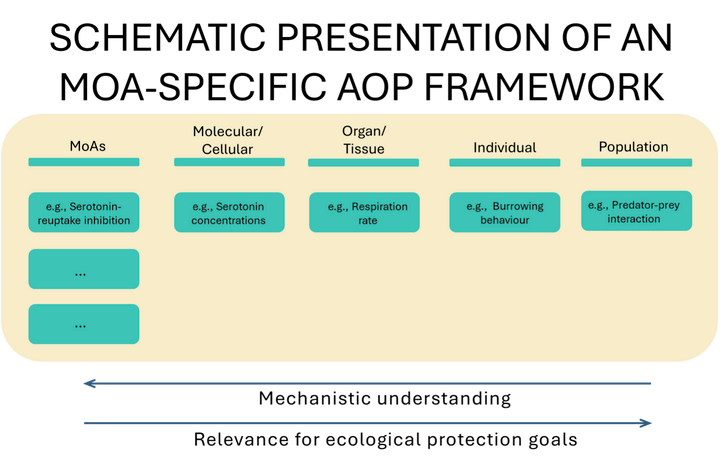
Figure 1: Schematic presentation of an MoA-specific AOP framework with the specific MoAs on the left (e.g., serotonin-reuptake inhibition) and endpoints related to this MoA suggested for the different biological levels (molecular/cellular, organ/tissue, individual and population).
Four Key Outputs of CHRONIC
- Non-conventional endpoints may be more sensitive than conventional endpoints, especially for specific MoAs (e.g., serotonin reuptake inhibitors).
- Even though sometimes more variable, these endpoints can provide important mechanistic insights into chemical MoAs.
- Non-conventional endpoints can reveal effects relevant for the population-level that are missed under current regulatory practices.
- AOPs should be better implemented in ERA practices by aligning non-conventional and conventional endpoints with chemical MoAs. Important lessons can also be learned from the incorporation of AOPs within human health risk assessment.
Food for Thought
To integrate the knowledge from CHRONIC and its final workshop into regulatory practice, several key actions were highlighted:
- Aligning non-conventional and conventional endpoints with AOPs will improve the understanding of chemical MoAs and hazards in ERA practices.
- One of the main challenges for the integration of non-conventional endpoints is that they are not standardized. There is a need for better reporting in scientific studies (e.g., CRED, EthoCRED) so that data meet regulatory reliability and relevance criteria. The availability of the underlying data should be FAIR, making it more easily accessible for regulators (e.g., the European Database for academic studies) and researchers.
- New scientific advances can and should be promoted by the active engagement of scientists in the regulatory process through participation in expert panels, guidance document development and OECD’s Standard Project Submission Form (SPSF).
- All available evidence needs to be considered when regulating chemicals under, for instance, REACH or the regulation for plant protection products. Non-conventional academic data often provide information on effects with population relevance that have been overlooked during chemical registration using conventional endpoints. Regulators should actively educate scientists on how to achieve reliability and relevance in ecotoxicological studies, thereby bridging the gap between regulators and scientists and enhancing the usefulness of academic data for regulatory actions.
Consensus is needed on how data are collected, reported and managed between the scientific community, business and regulatory bodies. From the scientific community, it is crucial to pay attention to the requirements of regulatory bodies and follow guidelines, focusing on detail and presenting all parameters that increase the repeatability and reproducibility of their studies, such as the OECD Guidance Document on the Generation, Reporting and Use of Research Data for Regulatory Assessments. If the scientific community fulfils the needs of regulators in terms of data accessibility, this will enable faster workflow and more informed regulatory decision-making.
Contact: [email protected]
